AquiSense
PearlSurface™ 24G9 UV-C LED Disinfection System
Introducing our flagship surface disinfection system, the PearlSurface™ 24G9 UV-C LED disinfection system offers laboratory-grade disinfection and sterilization in a self-contained unit.
The PearlSurface™ 24G9 offers consistent and reliable UV-C LED disinfection for a wide range of applications from medical equipment, personal items, industrial bottling, consumable surfaces, and even packing.


The PearlSurface™ 24G9
The new, easy and simple way to disinfect surfaces.
The PearlSurface™ 24G9 is made up of 3 x 3-inch square modules that can be operated individually for your disinfection needs or combined into large arrays to fit any application.
Each of the PearlSurface™ modules can be outfitted with a single wavelength and assembled in such a way as to provide multi-wavelength coverage. We have eliminated the threat of hotspots by homogenizing the LED’s light to output a uniform intensity distribution to treated surfaces.
You can select wavelengths for your specific needs between the range of 265nm and 285nm, allowing you to target and destroy particular bacteria. The PearlSurface™ is also able to select wavelengths outside this range, meaning it can handle any disinfection job you need.
UV-C LEDs allow for almost any wavelength to be selected, allowing for highly targeted pathogen reduction and destruction.
The PearlSurface is 1 of 3 new platforms
Benefits of the PearlSurface™
The PearlSurface™ UV-C LED disinfection system offers unrivaled benefits compared to mercury lamp alternatives.
1. Instant On/Off
One of the greatest benefits of the PearlSurface™ UV-C LED disinfection system is the instant on/off capabilities that UV-C LEDs offer. Waiting around for lamps to warm up and reach the desired wavelength for your disinfection needs no longer exists, with the PearlSurface™ you can quickly and instantly disinfect surfaces and equipment in a matter of seconds.
2. Low Power Consumption
Save money on your energy bills as the PearlSurface™ UV-C LED disinfection system is a cost-effective, low power consumption device. Making use of LEDs that require significantly less power than mercury lamp alternatives, the PearlSurface™ can operate for long periods of time for a fraction of the cost.
3. Lead-Free
When we came to design the PearlSurface™ we did not just want to improve disinfection performance, we wanted to create a disinfection product that was much safer for you to use.
The PearlSurface™ UV-C LED disinfection system contains zero lead, meaning it is safer and easier for you to use, without the need to worry about harmful lead contamination.
4. Mercury-Free
For so long, many industries had to rely on mercury lamps for surface disinfection and endured long warm-up times and high energy consumptions. Mercury is a harmful and dangerous element if not handled correctly and can cause serious harm.
The PearlSurface™ removes mercury altogether, using energy-efficient LEDs to produce UV-C light in a controlled and self-contained system. With instant disinfection capabilities and no waiting around for a harmful lamp to warm up, the PearlSurface™ is making mercury lamps a thing of the past.
5. No Chiller Needed
No chiller, no worries. The PearlSurface™ does not require a chiller to operate at full capacity, instead, the PearlSurface™ is an entirely self-contained modular system that is ready to go from the start to suit whatever your disinfection needs.
6. No Heat Transfer
Another drawback of conventional surface disinfection systems is the heat transfer that occurs to the surface and equipment that is being disinfected. Unaccountable heat transfer can be a costly and cumbersome issue that could lead to the surface and equipment becoming damaged and no longer fit for purpose.
The PearlSurface™ has zero heat transfer, meaning you no longer need to worry about damaging surfaces and equipment when disinfecting, allowing you to focus on the task at hand.
7. Selectable Wavelength
Another benefit of the PearlSurface™ UV-C LED disinfection system is the ability to select your desired wavelength and be instantly able to disinfect at that output. No waiting around and testing to see if you have the right wavelength and risking not fully destroying any bacteria or pathogen that might be present.
With different bacteria and pathogens requiring varying wavelengths to reduce and even destroy them, being able to easily and simply alter the wavelength saves you not only time and energy but gives you peace of mind.
8. Configurable Layout
One of the biggest benefits to the PearlSurface™ is the entirely modular system, meaning you can configure your PearlSurface™ for your specific needs. So whether you are tight on space and need a compact system or need to disinfect a lot of different equipment in a short time, the PearlSurface™ can be built to your needs.
What’s inside the PearlSurface™ 24G9
The PearlSurface™ 24G9 is made up of three core elements, the LED board, the conveyor disinfection array, and a custom disinfection array.
LED Board
The LED board the what makes the PearlSurface™ unique, our compact array of UV-C LEDs allow for pathogen reduction with the need for harmful and expensive chemical or mercury lamps.
Conveyor Disinfection Array
Our modular design means you can build your PearlSurface™ to your needs, with thermal management and all custom made to size, you can have a bespoke PearlSurface™ UV-C LED disinfection system.
Custom Disinfection Array
Whether you need a disinfection system that operates 24/7 or that functions to reduce a specific pathogen, our custom arrays can be fitted to suit your needs. From advanced disinfection to liquid cooling systems, all our PearlSurface™ models can be customized for your needs.
The Technology
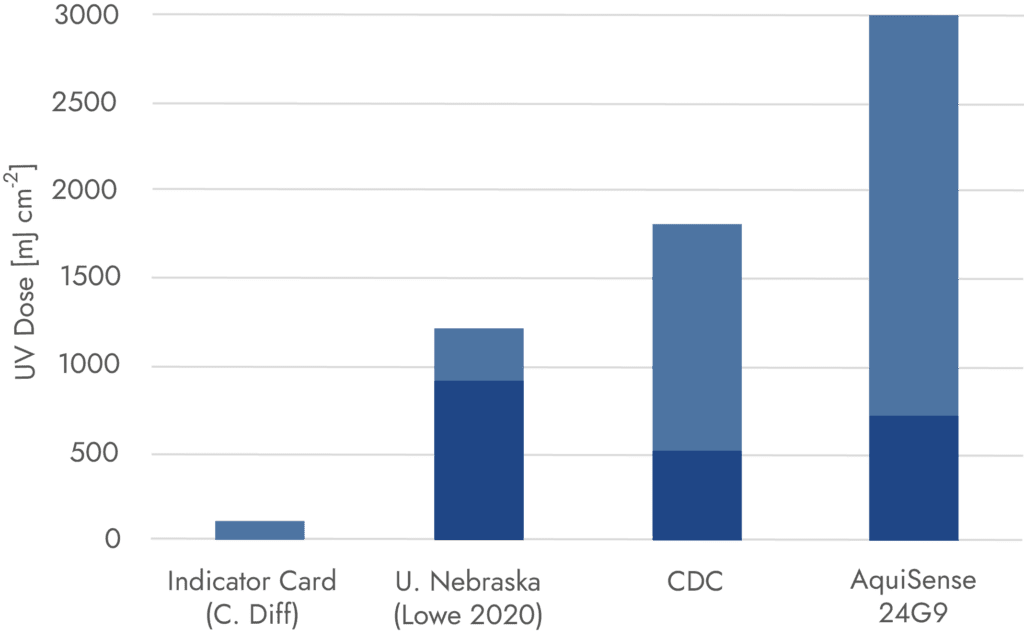
The PearlSurface 24G9 is supported by advanced optical designs using ray-tracing software. Our team used our experience in designing systems for high UV exposures up to 1,000 mJ cm-2 in under 20 minutes.
The CDC recommends a minimum UV Dose of 500 mJ/cm-2. AquiSense’s PearlSurface delivers a minimum of 700 mJ/cm-2 with 90% of the plane receiving a UV Dose over 1,000 and a maximum of 3,000 in parts of the plane.



Applications
The PearlSurface™ 24G9 has many applications and can be used in a range of industries and settings, including:
- Small healthcare clinics
- Hospital wards
- Nurse stations
- Medical offices
- Long term care facilities
- Laboratories
- First responders
- Dental offices
- Restaurants.
How safe is the PearlSurface™ 24G9?
The PearlSurface™ 24G9 is one the safest disinfection systems on the market. The PearlSurface™ 24G9 comes equipped with an integrated timer and a hinged door with safety interlock that prevents unwanted UV exposure outside the system.
The safety features are easy to use and ensure that the operator is safe when the device is active and ensuring it functions to the highest standards.
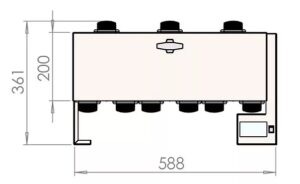
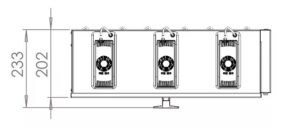
How big is the PearlSurface™ 24G9
Is this the right system for you, take a look at the dimensions below to see how the PearlSurface™ 24G9 could work for you. All measurements are in millimetres (mm):


About AquiSense
AquiSense is a leading UV-C LED product manufacturer developing laboratory research tools in addition to air, water, and surface treatment products. We operate as a business-to-business company supplying components to laboratories, Original Equipment Manufacturers (OEMs), and distributors within the manufacturing space.
Certification
The PearlAqua Deca has been certified by a 3rd party laboratory in accordance with US EPA drinking water guidelines.








